Physician Therapeutics
Sentra PM
Couldn't load pickup availability
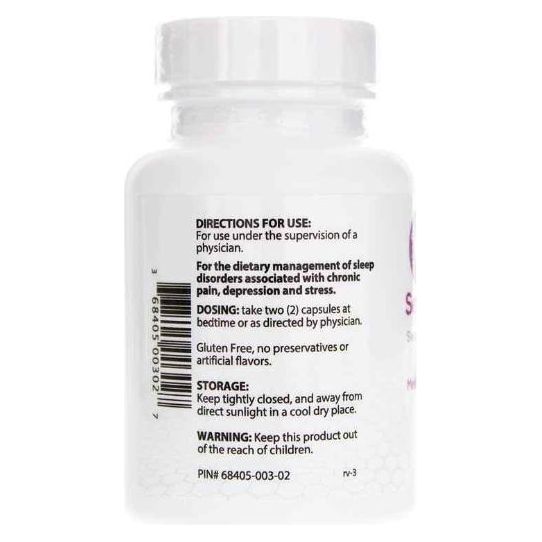
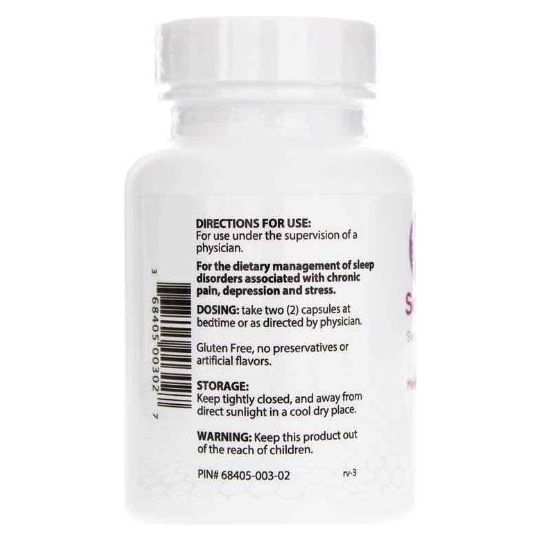
Sentra PM® for the dietary management of sleep disorders associated with depression and stress disorders. Sentra PM capsules by oral administration. A specially formulated Medical Food consisting of a proprietary formulation of amino acids and polyphenol ingredients in specific proportions for the nutritional management of the altered metabolic processes of sleep disorders (SD).
How Can Sentra PM Help?
Sentra PM provides the tools your body requires to fall asleep faster and stay asleep longer. Sentra PM addresses the increased metabolic requirements of sleep disorders and anxiety through a nutritional pathway, addressing specific amino acid deficiencies using a patented ingredient technology. Sentra PM works quickly and does not lose efficacy over time.
Is Sentra PM Right For Me?
Sentra PM is a non-addictive and effective way to reduce the time it takes to fall asleep. No severe side effects, significant morning grogginess, or the potential for addiction or tolerance build-up have been reported.
PRODUCT DESCRIPTION:
Sentra PM is a proprietary formulation of amino acids and other dietary factors to support induction, maintenance, and enhancement of the specific neurotransmitter activity involved in the physiology of SD. These ingredients fall into the classification of Generally Recognized as Safe (GRAS) as defined by the Food and Drug Administration (FDA) (Sections 201(s) and 409 of the Federal Food, Drug, and Cosmetic Act).
A GRAS substance is distinguished from a food additive on the basis of the common knowledge about the substance's safety for its intended use. The standard for an ingredient to achieve GRAS status requires a technical demonstration of non-toxicity and safety and general recognition of safety through widespread usage and agreement of that safety by experts in the field. The FDA determined Many ingredients to be GRAS and listed as such by regulation in Volume 21 Code of Federal Regulations (CFR) Sections 182, 184, and 186. Amino Acids Amino Acids are the building blocks of protein and are GRAS listed as humans have safely ingested them for thousands of years.
The formulations of the amino acids in Sentra PM are equivalent to those found in the usual human diet. Patients with sleep disorders may require more amino acids that cannot be obtained from a normal diet alone. Tryptophan, for example, is an essential amino acid. The body cannot make tryptophan and must obtain tryptophan from the diet. Tryptophan is needed to produce serotonin. Serotonin is required to induce sleep. Patients with sleep disorders have altered serotonin metabolism. Some patients with sleep disorders have a resistance to the use of tryptophan that is similar to the mechanism found in insulin resistance that is genetically determined. Patients with sleep disorders cannot acquire sufficient tryptophan from the diet to establish normal sleep architecture without ingesting a prohibitively large amount of calories, particularly calories from protein.
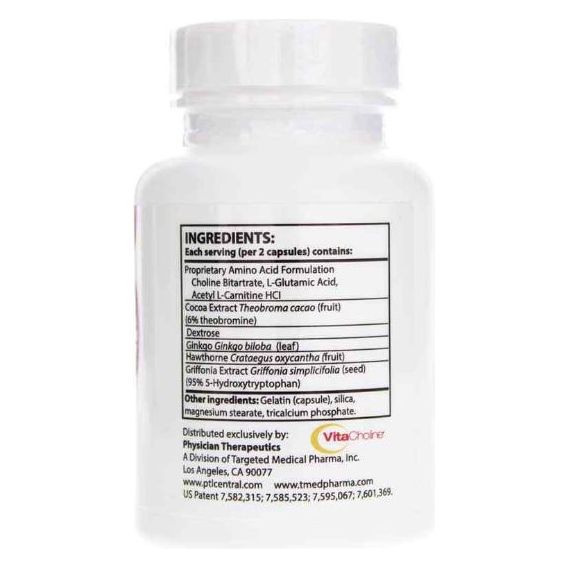
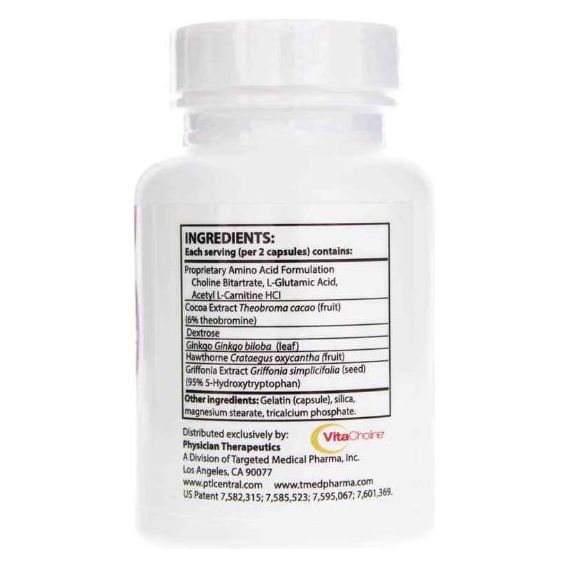
Other Ingredients Sentra PM contains the following “inactive” or other ingredients as fillers, excipients, and colorings: Gelatin, vegetable magnesium stearate, silicon dioxide, lac-resin, and carmine.
Share
Suggested Use
Suggested Use
one (1) to two (2) capsules of Sentra PM be taken at bedtime; Your doctor will determine the most appropriate dose for your condition. Patient must be under the ongoing supervision of a physician when taking Sentra PM.
Ingredients
Ingredients
Choline Bitartrate, Cocoa Extract, Glutamic Acid, Acetyl L-Carnitine HCL, Whey Protein Isolate (Milk), Dextrose, Ginkgo Biloba, Hawthorne Berry, Griffonia Seed
References
References
FAQ's
FAQ's
Shipping Policies
Shipping Policies
US Domestic Shipping:
Standard US Shipping Rates: *Saver Rate Shipping (3-5 Business Days)
US Domestic Order Subtotals of: $200.00 or more: FREE
At checkout, you will be prompted to choose a shipping method for your item(s). Shipping costs are dependent on the items in your order and the shipping method you select. Perishables and temperature-sensitive items, including suppositories, nasal sprays, and natural deodorants, require expedited shipping.
Note: Not all products sold at RevelationHealth.com qualify for free or discounted shipping.
Revelation Health LLC shipping hours are Monday through Friday 9AM to 5PM EST, excluding holidays. We do not process orders for shipping on Saturday or Sunday.
Product Returns:
You may return an un-opened, un-used, non-perishable* product within 15 days for a refund minus shipping cost and 20% restocking fee.
Please note: All sales are final and non-refundable when purchasing supplements from Systemic Formulas.
If you have opened the product and it is defective or contains ingredients we did not list on our site, you may also return it. Unfortunately, we cannot refund you if you do not like a product after trying it. There is too much variation in taste perception, digestion, and health status. What may work for one person may not work for another. We hope you understand.
To send back your unopened product, contact us via email to obtain an RMA number and an address to which the box should be sent. DO NOT SEND BACK PRODUCTS WITHOUT AN RMA#, or you will not get credit. Shipping the product back will be your responsibility. Once the product is returned, we will issue a refund, less the restocking fee and original shipping cost.
If you were shipped the wrong product by fault of ours, we will provide you with a free shipping label for its return (or you may be able to keep it) and we will credit you for that product as soon as we receive it back. The correct product will be shipped out to you as soon as we are informed of the error.
Perishables are Non-Refundable:
Returns are NOT possible on perishable items such as Protein Bars (i.e. Paleobars, Probiotics), suppositories, nasal sprays, or items that require temperature control.
Important Notice:
Perishables and temperature-sensitive items, including suppositories, nasal sprays, and natural deodorants, require expedited shipping. These packages may include ice packs for temperature control. You may purchase additional ices packs at check out or when prompted to add an ice pack.
To ensure product integrity, please retrieve your package promptly upon delivery and store it in a temperature-controlled environment as instructed on the packaging.
Generally, if you are shipping within the United States, you will have the option of upgrading your shipping method for faster delivery (Ground, Third Day, Second Day or Overnight Service*). If you choose to upgrade your shipping method, your order must be received and clear credit authorization by 12:00 p.m. (noon)** EST or your order may not be processed until the following business day. Business days are Monday-Friday, excluding observed holidays within the United States.
If you purchase the first-class mail option, Once your items are in the mail, the package is no longer the responsibility of Revelation Health LLC. We will not replace lost or stolen packages. If you purchased the Priority or Priority Express option, your package does come with standard insurance and we will file a claim for your lost or stolen package.
International Orders are not eligible for returns. Please check with Customs Regulations on importing supplements into your country. We are not liable or responsible for customs seizures. Taxes and Duty Fees are required prior to delivery.
UPS Next Day Air Services do NOT include Saturday delivery. Please call us to arrange for UPS Next Day Air Service Saturday delivery.
USPS Expedited and Priority Mail are NOT guaranteed services. Please choose UPS or FedEx for guaranteed services.
*Due to USPS zone restrictions, we cannot guarantee second-day or one to two day express delivery to all zones. **Does not guarantee Same-Day Shipping.
International Shipping:
International orders are not eligible for returns.
Important Notice Regarding International Shipments:
There are many country-specific laws, regulations, and restrictions that may impact our ability to ship your order and your ability to receive it. We recommend you contact your customs office to understand what regulations and restrictions may apply to orders shipping from the United States and/or the items you are ordering. Customs may delay your order or might reject it outright from being delivered based on local laws. In addition, you may incur higher than expected import duties or tariffs, your order may not be deliverable, or customs may quarantine or return your order back to us upon their inspection.
At this time we are unable to ship to addresses located in the following European Union and United Kingdom locations:
- Austria
- Belgium
- Bulgaria
- Croatia
- Cyprus
- Czechia
- Denmark
- England
- Estonia
- Finland
- France
- Germany
- Greece
- Hungary
- Ireland
- Italy
- Latvia
- Lithuania
- Luxembourg
- Malta
- Netherlands
- Northern Ireland
- Poland
- Portugal
- Romania
- Scotland
- Slovakia
- Slovenia
- Spain
- Sweden
- Wales
Any duties, tariffs or local taxes that may apply to your order are your responsibility. Revelation Health cannot anticipate or estimate country-specific duties, tariffs, and taxes that may apply to your order. It is your responsibility to research and understand these fees and pay them once levied. Our prices do not reflect any of these additional charges. These fees are determined by a local customs office and Revelation Health cannot be responsible for any such fees incurred.
Caution: If you are pregnant or nursing, consult with your healthcare practitioner before use. Keep out of reach of children.
*These statements have not been evaluated by the Food and Drug Administration. This product is not intended to diagnose, treat, cure or prevent any disease.
View full details
Recent Articles & News...
Free Shipping Bonus
Enjoy free shipping on US orders over $200
Fast Support
Feel free to let us know if you have any questions
Secure Payment
Safe & secure payment with encrypted checkout.
Subscribe To Our Newsletter
Get all the latest offers and product updates sent to your inbox
Follow us on: